Reference no: EM13554683
Uhura Resort opened for business on June 1 with eight air-conditioned units. Its trial balance on August 31 is as follows.
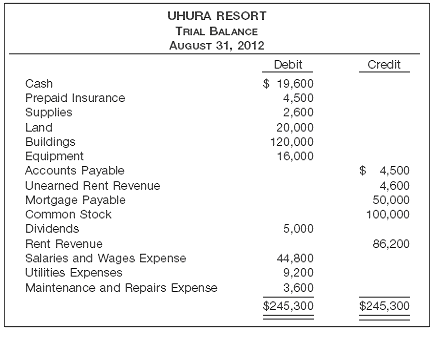
Other data:
1. The balance in prepaid insurance is a one-year premium paid on June 1, 2012.
2. An inventory count on August 31 shows $650 of supplies on hand.
3. Annual depreciation rates are buildings (4%) and equipment (10%). Salvage value is estimated to be 10% of cost.
4. Unearned Rent Revenue of $3,800 was earned prior to August 31.
5. Salaries of $375 were unpaid at August 31.
6. Rentals of $800 were due from tenants at August 31.
7. The mortgage interest rate is 8% per year.
Instructions
(a) Journalize the adjusting entries on August 31 for the 3-month period June 1-August 31. (Omit explanations.)
(b) Prepare an adjusted trial balance on August31.
|
Explain packing fraction for bcc lithium
: Determine the repeat distance, linear density, and Packing fraction for BCC lithium, which has a lattice parameter of .35089 nm, in the [100], [110], and [111] directions. Which of these directions is close packed
|
|
What are allowable departures from historical cost
: What are the allowable departures from historical Cost discussed in class? Two of them involve adjusting the cost to Market Value, which ones, what assets do each one adjust and how does handle unrealized gains?
|
|
Determine how much work does the engine do
: There are two answers required for this question. An engine operates between temperatures of 280 K and 700 K at 62.0 % of its maximum efficiency. how much work does the engine do
|
|
Why it is essential to wash the recrystallized material
: Why is it essential to wash the recrystallized material with cold, not warm or hot, solvent. What might occur if the wash with cold solvent was omitted
|
|
Prepare an adjusted trial balance on august31
: Journalize the adjusting entries on August 31 for the 3-month period June 1-August 31 and prepare an adjusted trial balance on August31.
|
|
Determine how much ice is required
: An aluminum container which has a mass of 0.350 kg holds 0.70 kg of water. The water and container are both at 45.0°C. How much ice is required
|
|
Estimate the frequency of siren perceived by the observer
: An ambulance is generating a siren sound at a frequency of 1,800 Hz. The velocity of sound is 335.0 m/s. What is the frequency of the siren perceived by the observer
|
|
Explain a mixture slowly during recrystallization
: Why is it important to use a minimum volume of hot solvent to dissolve an impure sample during recrystallization and why is it advisable to cool the solution of a mixture slowly during recrystallization
|
|
Evaluate the grams of nicl2 of the solution
: A 15.0% (w/w) NiCl2 (129.61 g/mol) solution has a density of 1.149 g/mL. Calculate the grams of NiCl2 contained in each liter of this solution
|