Reference no: EM131365896
Homework
1. Chapter 8: #3 (Note, there are no constraints for this problem)
2. Chapter 8: #9
3. Chapter 8: #21
4. Alex has money to invest in some combination of four investments (I1, I2, I3, and I4). There are three scenarios (A, B, and C) that may occur, and each investment will give a different return under each scenario (see below table, where the numbers indicate the percent return under each scenario). The probability of scenario A is 0.35, the probability of scenario B is 0.25, and the probability of scenario C is 0.4.
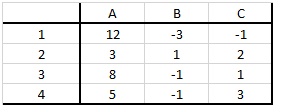
a) Formulate and solve the Markowitzian mean-variance problem of determining the portfolio which minimizes variance while maintaining an expected return of at least 2.5%.
b) Formulate and solve the Markowitzian mean-variance problem of determining the portfolio which maximizes expected return while maintaining a variance of no more than 30.
5. Three cleaning crews are available for a job that requires an area of 1000 square feet to be cleaned. Each cleaning crew charges a fixed cost that is only charged if the company is hired and a variable cost that is dependent on the number of square feet cleaned (see below). The job can be performed either by a single cleaner or by a combination of cleaners. Formulate and solve a mathematical programming model that will determine which crews to hire and how much (in square feet) of the job that each should do.
Company A Cost = 0.01x2 - 2x + 2000
Company B Cost = 0.005x2 + x + 4000
Company C Cost = 0.01x2 - x + 1000
|
Why are these two values so different
: Using the following data, calculate the pressure of 1.00mol of Ar(g) in a 40.0L container at 25 degrees Celcius using both the ideal gas equation and the real gas equation. Why are these two values so different? (aAr=136.3L2kPa mol-2; bAr=0.0322L ..
|
|
Do you agree with the viability of their policy
: When commenting to other students' posts, consider the following: Do you agree with the viability of their policy? Do you see any additional advantages or disadvantages to their policy
|
|
Sample of an ideal gas occupies
: A 4.00g sample of an ideal gas occupies 1.24L at 23 degrees Celcius and 1.12bar. What will be its volume at 14 degrees Celcius and 1.50bar?
|
|
Explain in detail one key aspect from one branch of buddhism
: Describe in detail *one* key aspect from one branch of Buddhism. How does this aspect differentiate this branch from other types of Buddhism?
|
|
Formulate and solve the markowitzian mean-variance problem
: OPR 330: Alex has money to invest in some combination of four investments (I1, I2, I3, and I4). There are three scenarios (A, B, and C) that may occur, and each investment will give a different return under each scenario (see below table, where th..
|
|
Determine the equilibrium partial pressures
: The reaction CH4(g) + H2O(g) CO(g) +H2(g) has Kc=8.73x10^2 at 400 degrees Celcius. Determine Kp then determine the equilibrium partial pressures AND the equilibrium concentrations of both the products if the 260 L container initially contained 3..
|
|
What type of leadership do you believe is required
: What type of leadership do you believe is required for groups that are comprised of individuals from various organizations, who recognize their own specific and different chains of command
|
|
Primary structure of a protein molecule
: What is meant by the primary structure of a protein molecule? How does it differ from the secondary structure of a protein? How are secondary protein structures held together? Discuss the importance of the tertiary and the quaternary structures of..
|
|
Why are you recommending the given five strategies
: Why are you recommending these five strategies? What are important considerations that the firm should be aware of when deciding on an enterprise network architecture?
|