Reversible and irreversible cells : A cell is said to be reversible if the following two conditions are fulfilled
(i) The chemical reaction of the cell dose not proceed further when an exactly equal external emf is applied.
(ii) Chemical reaction of the cell is reversed and current flows in opposite direction when external emf is slightly higher than that of cell. Any other cell, which does not obey the above two conditions, is termed as irreversible. The Daniell cell can be reversed but 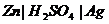 cell can not be irreversible in nature
cell can not be irreversible in nature
Email based Chemistry assignment help - homework help at Expertsmind
Are you searching chemistry expert for help with Reversible and irreversible cells questions? Reversible and irreversible cells topic is not easier to learn without external help? We at www.expertsmind.com offer finest service of Chemistry assignment help and chemistry homework help. Live tutors are available for 24x7 hours helping students in their Reversible and irreversible cells related problems. We provide step by step Reversible and irreversible cells question's answers with 100% plagiarism free content. We prepare quality content and notes for Reversible and irreversible cells topic under chemistry theory and study material. These are avail for subscribed users and they can get advantages anytime.
Why Expertsmind for assignment help
- Higher degree holder and experienced experts network
- Punctuality and responsibility of work
- Quality solution with 100% plagiarism free answers
- Time on Delivery
- Privacy of information and details
- Excellence in solving chemistry queries in excels and word format.
- Best tutoring assistance 24x7 hours