Chemistry Assignment Help >> f-block Elements Assignment Help
The f-block groups consist of those elements whose ions or atoms have valence electrons in f-orbitals. In fact electronic configurations may be slightly different from what is predicted by the Aufbau principle. The f-block elements are also known as inner transition elements, although that term is normally taken to include lawrencium and lutetium as well, which are part of the d-block elements. There are two series. Elements of the series where the electrons are in 5f orbitals belong to the actinide series. Elements of the series where the electrons are in 4f orbitals belong to the lanthanide series.
The elements form M3+ ions in the lanthanide series. In aqueous solution the lanthanides are surrounded by nine water molecules but the later lanthanides have a coordination number of 8. Cerium forms compounds with the oxidation state +4; Ce4+ has the very stable electronic configuration of the noble gas xenon. Ce(IV) is also a strong oxidising agent and Eu2+has the configuration [Xe]4f7 and is a strong reducing agent. The existence of Eu(II) is attributed to the stability of the half-filled f-shell.
The lighter actinides from uranium to americium show oxidation states of +3, +4, +5 and +6. The later actinides resemble the lanthanides in that the +3 oxidation state is favoured.
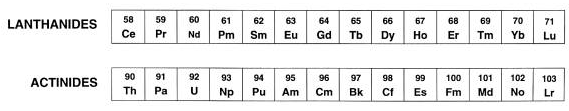
Lanthanides. (58Ce- 71Lu )
All lanthanides closely resemble lanthanum
Lanthanides are the elements in which the last electron enters into 4f-orbital
Lanthanoids are chemically similar to each other
Except for europium (III and II) and cerium (III and IV), the lanthanides occur as trivalent cations in nature.
Lanthanides are widely used in lasers mostly.
These elements deflect Infrared electromagnetic radiation and UV and are commonly used in the production of sunglass lenses.
The ionic radii of the lanthanoids decrease through the period — the so-called lanthanide contraction
Actinides ( 90Th– 103Lr)
They are radioactiveThese metals tarnish in air
Actinides are highly electro-positive ( show+3,+4,+5,+6 oxidation states)
Actinides react with boiling water or dilute acids to give H2 gas.
Actinides have number of isotopes.
They diThey directly combine with non-metals.
Similarities between Lanthanides and Actinides:
Both Actinides and lanthanides show +3 oxidation statesBoth are paramagnetic
They are electropositive and very reactive
Live Assistance with Qualified and Experienced Chemistry Experts: f-block Elements
We at
ExpertsMind have talented
chemistry experts who can help you in solving toughest chemistry problems in all topics. We offer chemistry assignment help, homework help, project help and instant online tutoring session anytime from anywhere. We provide quality of solution for assignments with step by step answers.