Reference no: EM131014999
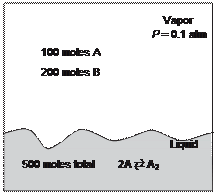
A vessel contains a liquid and a vapor phase in equilibrium at a pressure of 0.1 atm. The vapor phase contains 100 moles of species A and 200 moles of species B. The liquid phase contains 500 total moles of species. The saturation pressure of species A is 0.1 atm and of B is 0.5 atm. Both the vapor and the liquid phases behave ideally!
Species A dimerizes in the liquid phase and A2 is completely involatile.
(a) Calculate the equilibrium constant for the dimerization reaction.
(b) Calculate the values for the number of moles of A, B, and A2 in the liquid phase.
(c) A colleague maintains that the dimerization reaction does not occur in the liquid phase; rather, he believes that the liquid phase behaves nonideally. If species A occurs only as a monomer (as A, not A2) as he thinks, how many moles of species A would exist in the liquid phase [keep the total number of A atoms in the liquid phase the same as for part (b)]?
(d) Given your colleague's model in part (c), what value of gB is necessary for his model to fit the data?
(e) Can you explain nonideality in phase equilibria solely by introducing an association reaction to an ideal vapor-liquid system? Can an association reaction be used to explain negative 1 gB , 12 deviations from ideality?
|
Estimate the temperature of the furnace
: In addition to the species at the inlet, the outlet stream is measured to contain 0.40 mol/s of H2O vapor that has formed through chemical reaction. Assuming the reaction to produce water has reached equilibrium, estimate the temperature of the fu..
|
|
Contract limiting damages to a refund
: What damages should Mr. O'Connor receive? Would a refund of $125 be enough? What if the Tattoo Shoppe had a clause in its contract limiting damages to a refund? Would it be valid?
|
|
Complex divorce proceedings
: Tony Curtis, a respected actor, entered into a contract to write a novel for Doubleday & Company, publishers. Because of complex divorce proceedings and other per- sonal factors, Mr. Curtis was unable to submit a satis- factory manuscript. Doubled..
|
|
How many moles of hydrogen are produced
: If we build a reactor and run it under these conditions, could we ever get a lower conversion (less hydrogen) than that calculated in part (a)? Can we ever get a higher conversion? Explain.
|
|
What value of gb is necessary for his model to fit the data
: Can you explain nonideality in phase equilibria solely by introducing an association reaction to an ideal vapor-liquid system? Can an association reaction be used to explain negative 1 gB , 12 deviations from ideality?
|
|
Calculate the molar mass
: Demonstrate that acetonitrile is a -solvent for the polymer at 300K and calculate the molar mass.
|
|
What is its relationship to our understanding of classical
: Explain the culture and issues surrounding the HUAC and Hollywood-10 in America, film trends during the 1960s, the new wave of directors in the 1970s.
|
|
What is the balance in the cash account at the end of may
: 20. Required informationRefer to the information above. What is the balance in the Cash account at the end of May?$210,000.$390,000.$600,000.$810,000.
|
|
What is the maximum amount of h2
: The temperature is 1000 K and the pressure is 1000 Pa. You may assume that D hrxn does not change with temperature. What is the equilibrium constant at 298 K? What is the equilibrium constant at 1000 K?
|