Reference no: EM13164386
The Bottled Water Company Comprehensive Master Budget
In the following pages, you are given basic information for the operating budgets including the sales budget, the production budget, the direct materials purchases budget, the direct labor budget, the overhead budget, the selling and administrative expense budget, and the cost of goods manufactured budget. Complete the operating budgets on spreadsheet(s) in a workbook, using formulas and linking figures for maximum effectiveness of the spreadsheet.
Complete the budgeted income statement, linking the numbers from the supporting budgets to the income statement. Note, you are not given the information to complete the budgeted balance sheet, so a balance sheet is not expected to be a part of the assignment package.
Prepare an executive summary to Ginnie Adams, the owner of the Bottled Water Company, with your results from the comprehensive budget for the new product and how launching the new product would affect net income. Your spreadsheet(s) will be your attachment to the executive summary.
The project should include:
1. Executive Summary
2. Sales Budget
3. Production Budget
4. Direct Materials Budget
5. Direct Labor Budget
6. Overhead Budget
7. Selling and Administrative Expense Budget
8. Cost of Goods Manufactured Budget
9. Budget and Income Statement
Calculate the Bottled Water Company's net income for the new product in the coming year by completing the operating budgets and budgeted income state¬ment that follow.
I. Sales budget

2. Production budget:

3. Direct materials purchases budget:

4. Direct labor budget:

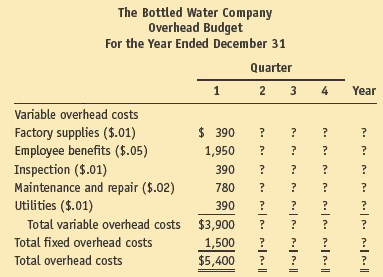
5. Overhead budget:
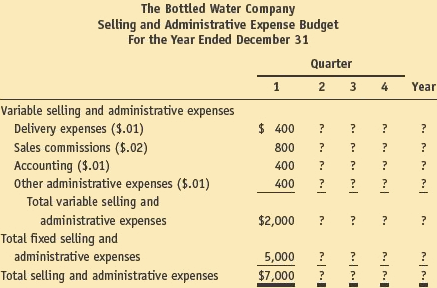
6. Selling and administrative expense budget:
7. Cost of goods manufactured budget:

8. Budgeted income statement

|
State using the average bond dissociation enthalpies
: Using the average bond dissociation enthalpies(BE) given, estimate the standard molar heat of reaction of the gas-phase reaction of hydrogen peroxide with methanol to yield formaldehyde and water:
|
|
Compute the solubility of ba
: Calculate the solubility of Ba(IO3)2 in 0.033 M solution of Mg(IO3)2 considering both the electolyte and common ion effects. Ba(IO3)2 Ksp
|
|
State ammonium chloride
: How many grams of ammonium chloride and how many milliliters 3.0 M sodium hydroxide should be added to 200 ml water and diluted to 500 ml to prepare a bufffer
|
|
Compute the percent manganese in the sample
: Calculate the percent manganese in the sample as Mn3O4 (only part or none of the manganese may exist in this form, but we can make the calculations on the assumption that it does)
|
|
Prepare an executive summary to ginnie adams
: Bottled Water Company Comprehensive Master Budget - Prepare an executive summary to Ginnie Adams, the owner of the Bottled Water Company, with your results from the comprehensive budget for the new product and how launching the new product would af..
|
|
Implement a simple 32 bit calculator program
: Operand A and Operand B have to be signed 32 bit integers. So an operand can be a negative number. For example, if you input is (-1 + 3), it should return 2. Operator must be one of the characters(' + ', ' - ', ' * ', ' / ', ' % ')
|
|
What is the concentration in the solution of the first ion
: what is the concentration in the solution of the first ion when the second one starts to precipitate at 25 C?
|
|
State naoh consuming one equivalent of base
: Compound A, C6H10O2, is optically active. It dissolves slowly in aqueous NaOH, consuming one equivalent of base. When the solution is acidified, A is slowly regenerated.
|
|
Calculate the partial pressure of each gas
: Assuming that the total pressure of the gases is 1.52 and that their mole ratio is 94 : 4.0 : 1.5 : 0.50, calculate the partial pressure (in atmospheres) of each gas.
|