Determine load carried by each cylinder:
A hollow steel cylinder of cross-sectional area 2000 mm2 concentrically surrounds a solid aluminium cylinder of cross-sectional area 6000 mm2. Both cylinders have the same length of 500 mm before a rigid block weighing 200 kN is applied at 20oC as shown in Figure. Determine
(a) The load carried by each cylinder at 60oC.
(b) The temperature rise required for the entire load to be carried by the aluminium cylinder alone.
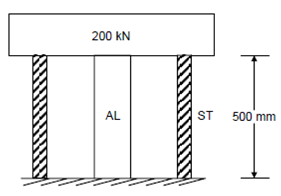
Figure
For computation purposes, take following values :
Esteel = 210 GN/m2 and Ealuminium = 70 GN/m2
σsteel = 12 × 10- 6 K-1 and αaluminium = 23 × 10- 6 K-1
Figure shows the free thermal expansions Δa and Δs together with the common expansion Δ under the load of 200 kN (the subscripts a and s standing for aluminium and steel respectively).
For a temperature rise of ΔT K,
We have,
Δa = 500 × 23 × 10-6 × ΔT = 11.5 × 10-3 ΔT mm
Δs = 500 × 12 × 10-6 × ΔT = 6 × 10-3 ΔT mm
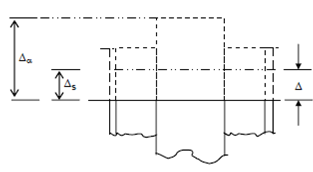
Under load, the strains are
εα = Δa -Δ /500
and εσ = Δs -Δ /500
and the corresponding stresses are as follows :
σα =70 × 103/500 (Δα - Δ) = 140 (Δα - Δ) N mm-2
σs = 210 × 103/500 (Δs - Δ) = 420 (Δs - Δ) N mm-2
For equilibrium of vertical forces,
σa × 6000 + σs × 2000 = 200 × 103 N
Substituting for σa, σs, Δa and Δs, we get
(11.5 × 10- 3 × ΔT - Δ) + (6 × 10-3 ΔT - Δ) = 5/21
Hence,
Δ= 8.75 × 10-3 ΔT - 5/42
The loads taken by the aluminium and the steel are therefore,
Pa = σa × 6000 N
= 840 ( 2.75 × 10-3 ΔT +5/42) kN
Ps = σs × 2000 N
= 840 ((5/42) - 2.75 × 10- 3 ΔT) kN
These equations will be valid as long as Δ is less than Δs. the load will be completely carried by aluminium when Δs becomes equal to Δ.
(a) at 60oC,
ΔT = 60 - 20 = 40 K
Pα = 840 ( 2.75 × 40 × 10-3 +5/42)
= 192.4 kN
Ps = 200 - 192.4 = 7.6 kN
(b) The load will be carried completely by aluminium when
6 × 10-3 × ΔT = 8.75 × 10- 3 × ΔT - 5/42
or ΔT = 5 × 103/2.75 × 42 = 43.3o C
i.e. at a temperature of (20 + 43.3) = 63.3oC.