Reference no: EM131243886
Foundations of Biomedical Science Exam
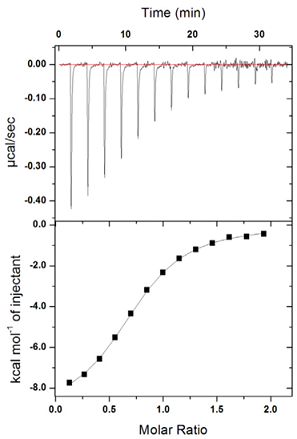
This figure represents an isothermal titration calorimetry experiment. Protein X and ligand Y interact inside a calorimeter chamber. The experiment is done like this:
- 200 μl of a 2 μM solution of protein X is placed inside the calorimeter chamber.
- A titration syringe is loaded with a solution containing Y (60 μM).
- At each point in time, as shown in the upper panel, 1 μl of the Y solution is injected into the calorimeter chamber. This injection results in the release of heat, as shown in the upper graph. The amount of heat being released changes with each injection because the binding sites are being saturated and there are fewer available sites to bind the ligand.
- Thus, the number of mols of X in the chamber remains constant, whereas the number of mols of Y increases with every injection.
- The experiment was done at 25°C
The heat released can be measured by the area of each one of the peaks shown in the upper panel. Calorimeters come with software that does this automatically for you and generate curves like that shown in the lower panel. The software corrects for the small dilution of X in the chamber (after 13 injections the volume has increased from 200 to 213 μl assuming that nothing gets removed). Therefore, the Molar Ratio (mols of Y/mol of X in the chamber) is an accurate number. The analysis software extracts several parameters from these data: ?H, ?S, K (equilibrium constant), and some estimate of the stoichiometry of the reaction.
Your job is to replace the instrument's software with your understanding of the analysis methodology. In other words, you must calculate the thermodynamic numbers associated with this reaction. You can do this using Excel or a piece of software available for download from the University (Prism). Because you need to plug in some numbers, there is a piece of freeware that is very useful for the digitation of plots (Plotdigitizer: https://plotdigitizer.sourceforge.net). If you don't want to use Plotdigitizer, you can always extract the data using a ruler and measuring distances to the X and Y axes.
These are your tasks:
1. Transform the lower graph into a classical binding isotherm and plot Bound [Y] vs Free [Y] (or Bound [Y] vs log Free [Y]).
2. Calculate the equilibrium constant and the binding stoichiometry.
3. If the stoichiometry is not 1, please discuss your result.
4. Calculate the ?H and the ?S of the reaction. The SIGNS are important.
|
Write an essay about medea and euripides
: Write an essay about Medea, Euripides, which addresses a specific question related to the social, cultural, political and/or Historical context, production history, or production challenges.
|
|
Discuss the anticipated impacts upon operating efficiencies
: Discuss the anticipated impacts (both positive and negative) upon operating efficiencies, and recommend solutions to minimize the negative impacts.
|
|
What is your vision of an open and just society
: As individuals, how do we promote inclusion, respect for diversity, and support for national and global initiatives dedicated to eliminating disparities and creating open, healthy societies?
|
|
What is your professional passion and what do you believe
: In a 500-word essay, write a statement of personal and professional belief. Answer the questions: "What motivates you. What's your professional passion? What do you believe?"
|
|
Find the equilibrium constant and the binding stoichiometry
: These are your tasks: Transform the lower graph into a classical binding isotherm and plot Bound [Y] vs Free [Y] (or Bound [Y] vs log Free [Y]). Calculate the equilibrium constant and the binding stoichiometry
|
|
Examine the selected companys specific practices or policies
: Examine the selected company's specific practices or policies. Speculate on the major influences that these practices or policies have on individual and organizational outcomes.
|
|
What is the information systems description framework
: You to use the Information Systems Description Framework (ISDF) to describe an information system that you choose from your own world (e.g. from your work, home, community, or elsewhere) or a new information system you are proposing.
|
|
Recast the income statement in multi step form
: Recast the income statement in multi- step form, including allocating income taxes to appropriate items and showing earnings per share figures.
|
|
Critical points-points of inflection
: Sketch the graph of f(x) = 4x^3 - x^4, clearly indicating any critical points, points of inflection, intervals on which f is increasing or de- creasing, intervals on which f is concave up or concave down and x- and y-intercepts.
|