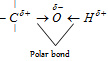
Chemical properties of monohydric alcohols : Characteristic reaction of alcohol are the reaction of the - OH group. The reactions of the hydroxyl group consists of either cleavage of C - O bond or the cleavage of O - H bond.
C - O bond is weaker in the case of tertiary alcohols due to +I effect of alkyl groups while - OH bond is weaker in primary alcohols as electron density increase between O - H bond and hydrogen tends to separates as a proton.
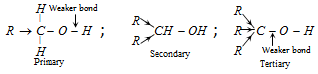
Thus primary alcohols give the most of reaction by cleavage of O - H bond while tertiary alcohols are most reactive because of cleavage of C - O bond. Hence - O - H cleavage reactivity order : Primary > Secondary > Tertiary and C - O - cleavage reactivity order : Tertiary > Secondary > Primary alcohol
(i) Alkylation : 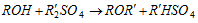
(ii) Reaction involving cleavage of 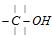 with removal or substitution of -OH group
with removal or substitution of -OH group
(a) Reaction with hydrogen halides : Alcohols give alkyl halide. The reactivity of HX is in the order of HI > HBr > HCl and the reactivity of ROH is in the order of benzyl > allyl > 3° > 2°> 1°. The reaction follows a nucleophilic substitution mechanism.
Grove's process
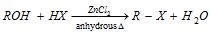
If alcohols react with HI and red phosphorus, alkane will be formed.
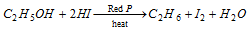
Primary alcohols follow S N2 mechanism .
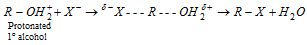
In secondary and tertiary alcohols, the S N1 mechanism operates
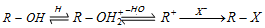
(b) Reaction with PCl5 : 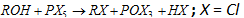 (Analytical test for alcohols)
(Analytical test for alcohols)
(c) Reaction with PCl3 :
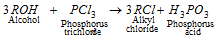
(d) Reaction with thionyl chloride [SOCl2] :
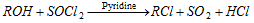
(e) Reaction with ammonia :
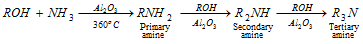
(f) Reaction with HNO3 :
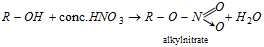
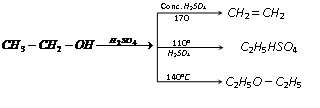
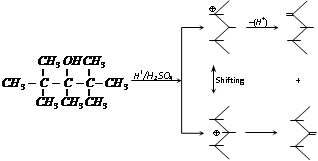
(g) Reaction with H2SO4 [Dehydration of alcohol] : The elimination of water from a compound is known as dehydration. The sequence of simple dehydration is Tertiary > Secondary > primary alcohol. The multiplication of dehydration of alcohols is depending upon the nature of dehydrating agents and temperature.
Email based Chemistry assignment help - homework help at Expertsmind
Are you searching chemistry expert for help with Chemical properties of monohydric alcohols questions? Chemical properties of monohydric alcohols topic is not easier to learn without external help? We at www.expertsmind.com offer finest service of Chemistry assignment help and chemistry homework help. Live tutors are available for 24x7 hours helping students in their Chemical properties of monohydric alcohols related problems. We provide step by step Chemical properties of monohydric alcohols question's answers with 100% plagiarism free content. We prepare quality content and notes for Chemical properties of monohydric alcohols topic under chemistry theory and study material. These are avail for subscribed users and they can get advantages anytime.
Why Expertsmind for assignment help
- Higher degree holder and experienced experts network
- Punctuality and responsibility of work
- Quality solution with 100% plagiarism free answers
- Time on Delivery
- Privacy of information and details
- Excellence in solving chemistry queries in excels and word format.
- Best tutoring assistance 24x7 hours